Atomic theory scientists4/2/2024 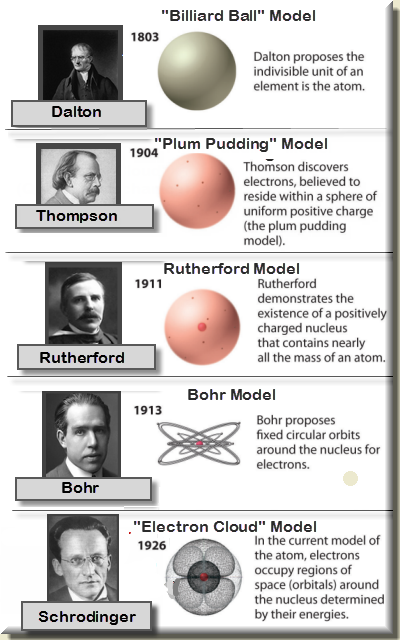
The properties of different elements are respective of the properties of the atoms present in the elements.In a given compound, the number and kinds of atoms are constant.The mass of elements combining during a chemical process indicates the total mass of atoms combined during the process.During a chemical combination process, the combination of two elements occurs by the union of the elements in a simple whole-number ratio to produce a compound atom (molecule).The atoms of different elements, however, are different and have different mass as well as other properties. Atoms are specific to elements, and all the atoms of a specific element are similar in all aspects and also have the same mass.This is called the principle of the indestructibility of matter. These particles can also not transform into the atoms of other elements. Atoms follow the law of conservation of mass and can neither be created nor destroyed during chemical processes.Atoms are the most basic unit of a chemical species that do not undergo any division during a chemical reaction.All matters are composed of tiny, indivisible, and discrete particles of units called atoms.The following are the main postulates of Dalton’s Atomic Theory The most essential concept of Dalton’s atomic theory is the suggestion that all matters are composed of atoms, but there are other different concepts in the form of postulates of the theory.In modern chemistry, Dalton’s atomic theory has been modified due to some of the drawbacks in the principle.Dalton, in the atomic theory, also introduced the word atom as the most fundamental particle of a chemical substance.Dalton’s atomic theory is based on two other important chemical laws of the time the Law of conservation of mass by Antoine Lavoisier and the Law of definite proportions by Joseph Proust. 
The atomic theory was developed by John Daltons after he made the observation that chemical substances combine and break down into other substances by weight which suggested that such chemical substances are made of smaller particles with a particular weight.Dalton’s atomic theory is a scientific concept which states that all matters are composed of tiny indivisible and discrete particles called atoms.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
